Related Post
2026 Expert Guide| Ultrasonicator for Chromatin Shearing Made Easy
2026-01-15Ultrasonicator for Chromatin Shearing is a precision instrument for controlled chromatin fragmentation. It applies focused ultrasound to deliver consistent fragment sizes for ChIP-seq and NGS. It reduces variability and protects biological signals. In this expert guide, we outline essential settings and safeguards. What matters most is not obvious.
What Is a Ultrasonicator for Chromatin Shearing in Detail
An Ultrasonicator for Chromatin Shearing applies focused high-frequency sound waves to break chromatin into controlled fragment sizes, typically in the 150 - 300 bp range for ChIP-seq. This method preserves protein - DNA interactions while delivering reproducible fragment distributions.
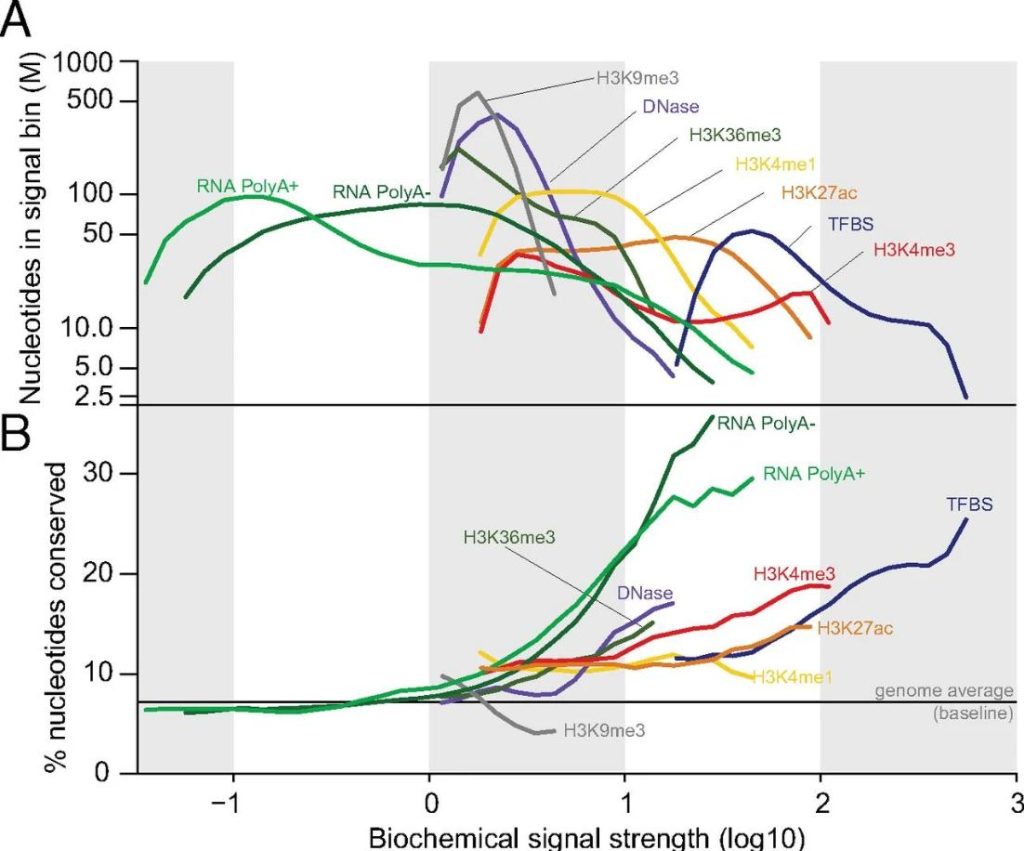
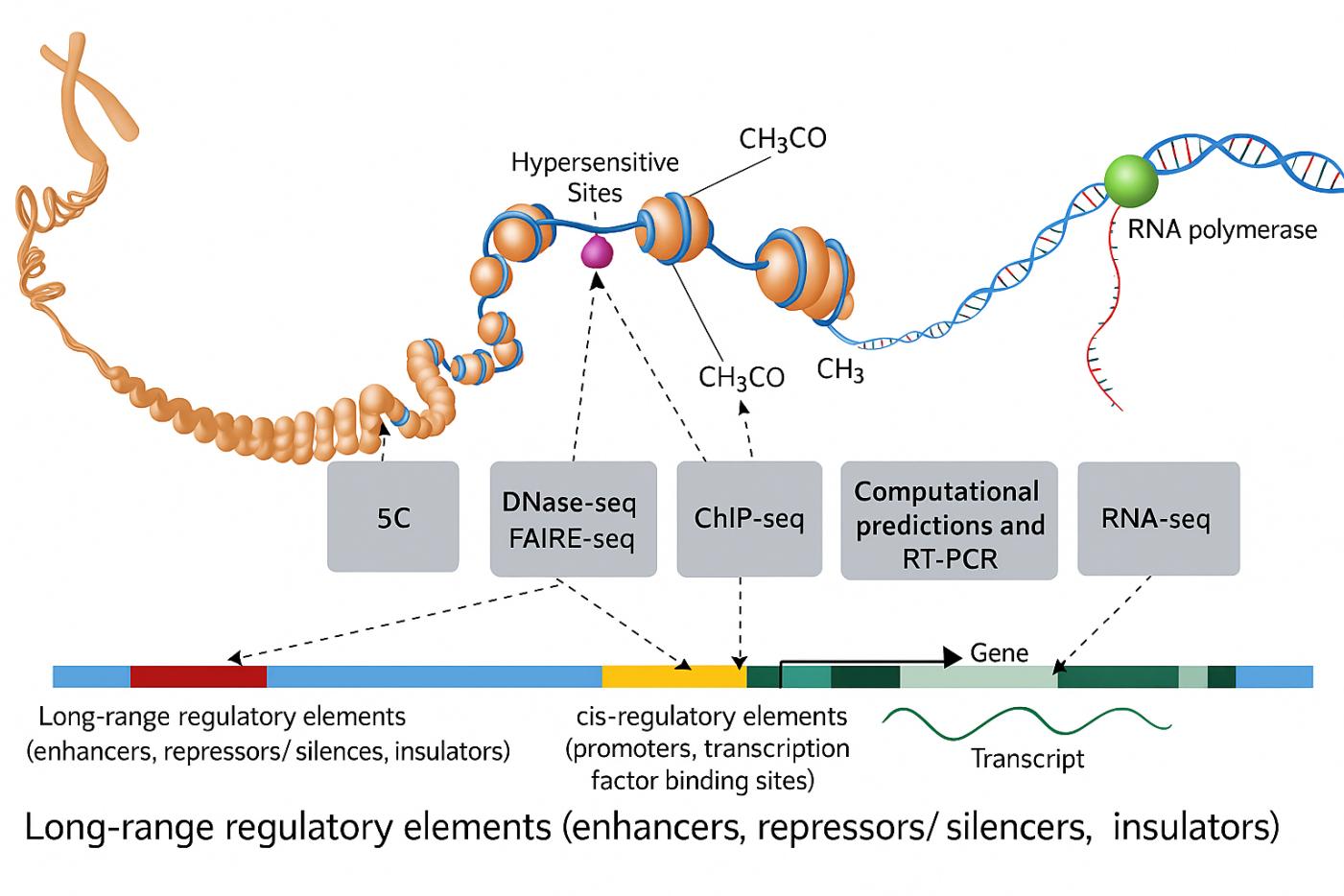
(Defining functional DNA elements in the human genome | PNAS)
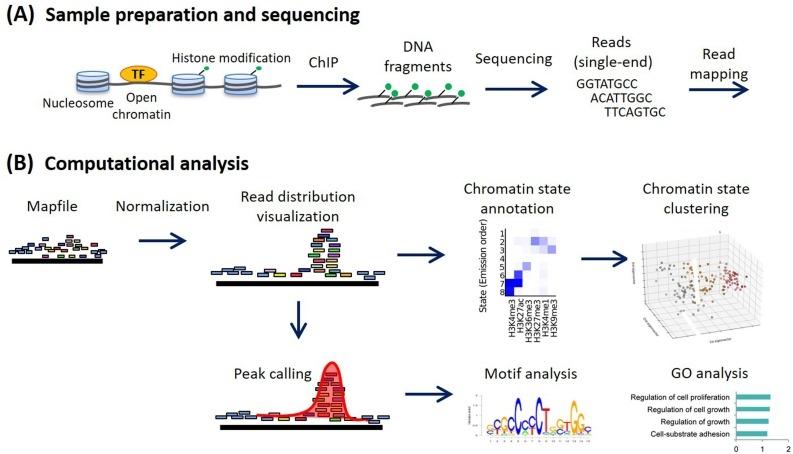
Pioneering studies from Michael Snyder, Barbara Wold, Bradley E. Bernstein, and Bing Ren helped define modern ChIP-seq practice. Their teams showed that careful sonication is central to signal-to-noise, peak resolution, and cross-tudy comparability. As genomics scaled through ENCODE and related consortia, standardized sonication became a core requirement.
Today's focused ultrasonicators use confocal acoustic technology to concentrate energy exactly where it is needed. They combine non-contact energy transfer, true low-temperature control, and automated cooling to reduce human error and deliver consistent results across users, instruments, and sites.
- Why Labs Struggle With Chromatin Shearing?
Chromatin shearing sits at a critical junction in ChIP-seq, ATAC-related workflows, and broad genomics assays. If you're seeing:
•Inconsistent results from manual sonication and fluctuating power
•Heat damage to protein - DNA complexes and skewed binding profiles
•Contamination from contact probes or shared baths
•Outcomes that vary by operator and schedule
•Noise that disrupts shared lab spaces
•External cooling and PCs taking over your bench
Switch to a modern ultrasonicator with precise acoustic focus, reliable low-temp control, non-contact energy, and automated cooling - so your shearing is consistent, quiet, and easy to reproduce.
How It Streamlines Workflow (Step By Step)
Step 1: Prepare Samples and Controls
Standardized preparation establishes predictable inputs. Paired controls provide variance benchmarking. Consistent volume handling supports cross-run comparability. Equal aliquots help stabilize outcomes. Uniform dispensing curbs operator-to-operator variability. Control checks confirm the desired fragment profile. Fragment distributions are calibrated to downstream performance targets.
Step 2: Load the Vessel and Establish Acoustic Coupling
The instrument's non-contact holder positions tubes for precise coupling. Focused acoustics enter the sample without a probe interface. This reduces contamination pathways and removes probe maintenance. Confocal energy delivery emphasizes the sample core over vessel walls.
Step 3: Select a Validated Method
Validated programs reflect the sample type and fragment intent. Methods encapsulate key settings such as burst dynamics, cycle count, and duty ratio. Standardized methods cut human error and speed training. If needed, select the advanced panel to fine-tune parameters for high-salt buffers or viscous lysates.
Step 4: Engage True Low-Temperature Control
Activate constant-temperature processing. The instrument's high-sensitivity sensors monitor the sample zone rather than ambient air. Automated cooling holds the target temperature, typically in the 4 - 8°C band for proteo - DNA complex protection. This prevents heat spikes that can weaken protein - DNA binding or modify epitopes.
Step 5: Auto-Cooling Pre-Run Check
Initiate a brief pre-chill. The built-in semiconductor refrigeration system reaches setpoint quickly. Unlike external chillers, the integrated unit eliminates hoses, condensate, and footprint. A stable temperature baseline improves fragmentation uniformity across cycles and batches.
Step 6: Start the Run With Real-Time Safeguards
Click run. Focused ultrasound energy is delivered in short, controlled bursts. Real-time feedback adjusts duty and pause intervals if temperature drifts. No-contact delivery produces uniform cavitation in the sample volume. Short and punchy: Confocal focusing = less stray energy, consistent sample intensity, zero container resonance.
Step 7: Monitor and Standardize
During processing, the system logs temperature, cycle counts, and energy metrics. Operators see visual status without noise distraction, thanks to quiet operation. If any deviation appears, automated safeguards correct it. This reduces operator bias and standardizes outcomes across shifts and sites.
Step 8: End-Run Cooling and Hold
When the method completes, auto-cooling engages immediately. Rapid cooling fixes fragment distributions and prevents thermal stress post-run. Samples remain at setpoint while you prepare for subsequent steps like reverse cross-linking or cleanup.
Step 9: Verify Fragmentation Quality
Remove a small aliquot for QC. Assess the distribution with capillary electrophoresis or gel imaging. If the profile skews high or low, adjust cycle count or burst duration in the method. Because parameters and temperature are logged, adjustments are straightforward and reproducible.
Step 10: Scale and Automate
Save the optimized method. Apply it across replicates or new projects. Integrated design and simple controls minimize setup time, while constant-temperature processing and non-contact energy deliver the same results batch after batch. This stepwise approach standardizes shearing, lowers human error, and strengthens cross-study comparability.
Together, these steps demonstrate how an Ultrasonicator for Chromatin Shearing unites low-temperature assurance, non-contact integrity, and auto-cooling control to streamline complex workflows. The result is consistent fragment size, protected protein - DNA complexes, and reproducible outcomes suitable for high-throughput ChIP-seq, ATAC-seq preprocessing, and broader NGS library preparation.
Key Features and Applications
- Focused confocal acoustics for accurate energy delivery
- Non-contact sample processing to minimize contamination risk
- True low-temperature, constant-temperature control in the sample zone
- Built-in high-efficiency semiconductor refrigeration for fast cooling
- Quiet operation, no sound enclosure required
- Integrated design without external computer or chiller, saving bench space
- Simple parameters and click-to-run methods for rapid training

Applications span DNA, RNA, and chromatin shearing, FFPE deparaffinization workflows, genome fragmentation for NGS library preparation, cell and tissue disruption for nucleic acid or protein extraction, and general tissue homogenization. Controlled fragmentation enhances ChIP-seq peak sharpness and cuts variability, making it easier to detect authentic biological signals.
Longlight Solutions and Call-to-Action
Longlight's end-to-end genomics solutions fit your Ultrasonicator for Chromatin Shearing from day one. We provide focused ultrasonication instruments, validated workflows, and high-grade reagents and consumables - including precast agarose gels for rapid QC, nucleic acid scavengers, Qubit tubes for dependable quantification, extraction kits, and NGS-optimized library prep kits.
Efficiency with accuracy - built in. We standardize chromatin shearing, target fragment sizes to fit your assays, and align methods with kits to accelerate results.
Call to Action: Contact Longlight for a consult or demo, or request a starter consumables set for ChIP-seq or NGS. Add low-temperature, no-contact, auto-cooled shearing to your lab for consistent performance.