Related Post
Cryo-EM: Visualizing Biomolecules at Near-Atomic Resolution
2026-02-21Cryo-EM: Visualizing Biomolecules at Near-Atomic Resolution is reshaping structural biology because it helps researchers observe proteins, complexes, and viruses in conditions that are closer to real life—without demanding crystals or overly aggressive sample handling. Longlight Technology supports cryo-electron microscopy projects with clear workflows, transparent deliverables, and practical guidance that helps teams move from “a promising sample” to “a defensible structure” faster and with fewer dead ends.
Scientists break resolution records to visualize individual atoms with single-particle cryo-EM
Instead of treating cryo-EM as a mysterious, high-end service that only works for perfect samples, we approach it like a structured decision system: screen what you have, learn what it is doing, and only scale up to high-resolution data collection when the sample is truly ready. That mindset saves time, budget, and precious material—especially for first-time users who need results that can survive internal review and peer review.
Why Cryo-EM Matters for Modern Structural Biology
What is Cryo-EM
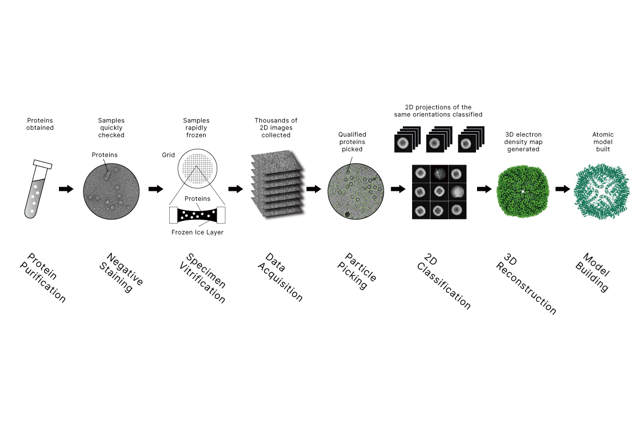
Cryo-EM (cryo–electron microscopy) is a way to “see” biological molecules by imaging them with an electron microscope after rapidly freezing them in a thin layer of vitreous (glass-like) ice. Freezing locks molecules in a near-native state without chemical fixation or crystallization, and computers combine many 2D views to reconstruct a 3D structure—often at near-atomic resolution for well-behaved samples.
How Cryo-EM Works (Simple View)
•Prepare sample (protein, complex, virus, etc.) in solution
•Vitrify it by rapid freezing so water becomes glass-like ice (no ice crystals)
•Image thousands to millions of particles using a transmission electron microscope
•Compute: align, classify, and reconstruct a 3D map; sometimes build an atomic model
Main Cryo-EM Methods
•Single-Particle Analysis (SPA): best for purified proteins/complexes; highest resolutions are common here.
•Cryo-Electron Tomography (Cryo-ET): 3D imaging of structures in cells or native environments; great for spatial context.
•Micro-Electron Diffraction (MicroED): for very small crystals (nano/microcrystals) when crystallography is hard.
Why People Choose Cryo-EM
•No need to grow crystals
•Captures molecules closer to their native state
•Works well for large complexes and many “difficult” targets
•Can reveal multiple structural states (motion/flexibility)
In life science, the fastest way to understand a biomolecule is often to see its shape and how that shape changes. Cryo-EM makes this possible for targets that are difficult, flexible, or unstable—exactly the targets many teams care about most. Cryogenic electron microscopy builds on transmission electron microscopy and can reconstruct 3D structures from sub-nanometer to near-atomic resolution, depending on sample behavior and data quality.
This is especially valuable in high-impact areas where details drive decisions:
•Drug discovery: binding pockets, interface geometry, and induced-fit changes
•Antibodies and vaccines: epitope mapping, neutralization mechanisms, and complex stability
•Virology: capsid organization, conformational shifts, and assembly pathways
•Membrane proteins: channels, transporters, receptors, and multi-pass complexes
Many modern targets do not crystallize easily, and some never will. Cryo-EM often converts “this is too hard to crystallize” into “this is testable now,” because the workflow is built for iteration: you screen quickly, adjust conditions intelligently, and then scale up once the particles behave.
Cryo-EM Vs X-Ray Crystallography: What Beginners Should Know
X-ray crystallography remains a powerful method and can reach extremely high resolution in the right case. But if you are new to structural biology project planning, it helps to focus on risk and probability, not just theoretical maximum resolution.
Cryo-EM reduces several common project blockers:
•No crystallization required, which removes a major uncertainty
•Near-native state preserved in vitrified ice, improving biological relevance
•Flexible or dynamic targets can be evaluated rather than “averaged away”
•Hard targets become more feasible (membrane proteins, large assemblies, viruses)
•Heterogeneity can sometimes be separated computationally into distinct states
•Lower purity tolerance may be possible for early discovery samples (case-dependent)
•Less sample waste compared to repeated crystallization screening
A simple way to think about it: crystallography can be extraordinary when crystallization works, but cryo-EM is often the more predictable route for difficult targets, multi-component complexes, and projects where you cannot afford months of trial-and-error.
Comparison of X-ray Crystallography, NMR and EM - Creative Biostructure
Structural Solutions We Provide: From Screening to Near-Atomic Models
At Longlight Technology, our cryo-EM service structure matches how real projects progress. We group work into three practical goals—so your pathway fits your sample and the question you are trying to answer.
Sample Suitability Assessment With Negative Stain Screening
Before investing in cryogenic grids and high-end microscope time, negative stain screening acts as a reality check. It is typically done at room temperature and helps answer the first question that matters: does the sample behave like a structural sample?
Negative stain can reveal:
•aggregation or clumping
•particle integrity and morphology
•size distribution and concentration suitability
•gross heterogeneity (too many “shapes” at once)
•signs of instability or degradation
Think of negative stain as a quality gate. It is not meant to publish high-resolution maps. It is meant to prevent expensive data collection on a sample that is not ready—and to guide the next optimization step with evidence instead of guesses.
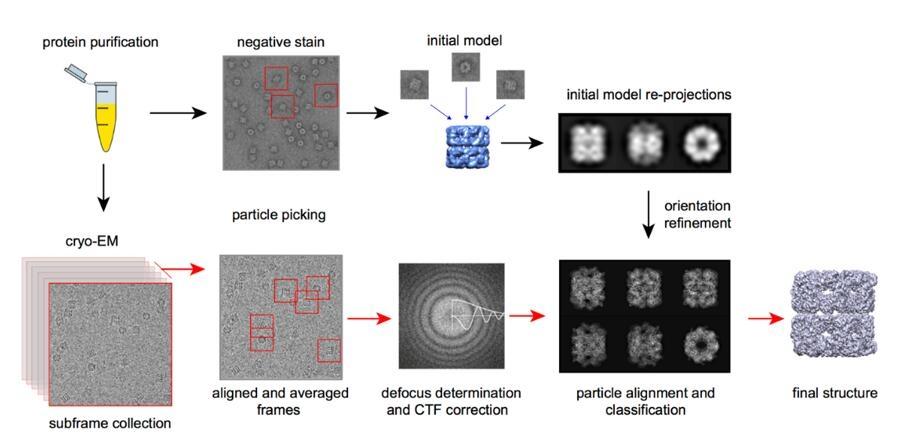
High-Resolution Structure Determination With Single Particle Analysis
For many soluble proteins and complexes, single particle analysis (SPA) is the most common path to high-resolution results. Large datasets contain individual particle views from many orientations. Computational processing aligns and classifies these particle images and reconstructs a 3D density map. When the map supports it, an atomic model can be built and refined to reveal mechanisms that are invisible to biochemical readouts alone.
SPA is where Cryo-EM: Visualizing Biomolecules at Near-Atomic Resolution becomes directly actionable: binding interfaces, domain movement, and structural explanations for activity, inhibition, or specificity can be described with confidence.
In Situ Structural Analysis With Cryo-Electron Tomography
If your question is not “what does this purified particle look like,” but rather “how is it organized in context,” cryo-electron tomography (Cryo-ET) is a better fit. Tomography can visualize structures in a more native environment, which is useful for:
•membrane-associated assemblies
•large cellular complexes
•virus-host interaction organization
•spatial arrangement and architecture questions
Cryo-ET is often chosen when the spatial story matters as much as the molecular shape.
Our Service Workflow and What You Receive
A cryo-EM project should not feel like a black box. We build clarity into each stage so you always know what is happening, what was observed, and what decisions are being made.
Typical workflow:
Project Consultation → NDA Signing → Service Agreement Confirmation → Sample Receiving → Quality Inspection → Negative Stain Screening → Cryo-EM Data Collection → Data Processing → Report Delivery
Deliverables are structured for real research use, not just for “a nice picture.” Depending on scope, you can receive:
•Raw cryo-EM movies (with gain reference files where applicable)
•Key intermediate processing outputs
•Final 3D density maps with resolution and quality metrics
•Atomic coordinate models (when the density supports model building)
•Validation and cross-check reporting (for example, structural quality assessments)
•Organized data delivery via secure cloud or portable storage
The goal is simple: you should be able to reproduce the work, reprocess if needed, and publish with confidence—without chasing files later or wondering how conclusions were reached.
Equipment Platforms That Match Different Project Stages
Different samples require different levels of instrument power and strategy. Paying for a flagship system to answer a basic screening question is inefficient, and it often slows projects down.
Our cryo-EM service capability is supported by platforms that cover screening through high-resolution structure determination, such as:
•Talos L120C G2: efficient TEM/cryo-EM screening and evaluation
•Glacios 2 (200 kV): strong workhorse system for routine SPA and cryo-ET workflows
•Titan Krios G4 (300 kV): flagship platform designed for stability, automation, throughput, and top-tier resolution potential
This tiered approach supports a practical principle: use the right microscope at the right moment. Screening stays efficient, and high-resolution collection happens only when the sample is ready to justify it.
Practical Project Requirements, Delivery Windows, and a Clear Next Step
If you are planning your first structural project, sample preparation details matter. Most delays are not caused by “the microscope,” but by avoidable sample issues such as instability, aggregation, or incompatible buffer components.
Typical minimum sample guidance:
•Negative stain: ~1 g/L, ~100 µL
•SPA (soluble proteins): ~1 g/L, ~100 µL
•SPA (membrane proteins): ~1 g/L, ~100 µL (often adjustable by discussion)
Typical buffer guidance:
•pH 6.0–8.5
•Salt concentration <200 mM
•Prefer low glycerol and low azide (case-by-case optimization is possible)
Typical delivery windows:
•Negative stain screening: 1–2 weeks
•SPA preliminary outcomes: 6–8 weeks
•SPA high-resolution model (when achievable): ~2–3 months
CTA: If you want to de-risk a new target quickly, start with negative stain screening. Send Longlight Technology your sample details (target type, buffer, concentration, and estimated purity), and we will recommend the most efficient route—negative stain, SPA, or cryo-ET—based on your scientific goal and timeline.
Finally, many cryo-EM studies connect to upstream and downstream biology. Longlight Technology also supports broader molecular biology and genomics workflows, including NGS-related instruments (such as focused ultrasonication) and commonly used consumables and kits (precast agarose gels, nucleic acid extraction kits, and library preparation kits), so your structural work can link smoothly to discovery, validation, and publication-ready study design.